A new study shows a clear association between the prophylactic use of five-layer foam sacral dressings and reductions in pressure injury rates. Specifically, the study looked at the prophylactic use of Mölnlycke’s Mepilex® Border Sacrum dressing in the acute care setting over a six-year period (2010-2015). (more…)
Read MoreTag: wound care
Three registered nurses pass National Wound Care course
Three registered nurses (RN) at Panhandle Home Health have passed the National Wound Care & Ostomy Certification Course & Exam (WCC) as part of Panhandle Home Health’s Wound Care Initiative, started in 2013 with a single WCC-certified RN, Cathy Reifer. In 2015, WISH (Women Investing in Shepherd) awarded their inaugural grant to a regional nonprofit, Panhandle Home Health. This grant of $26,250, along with additional private donations and grants, has allowed thirteen nurses to participate in the intensive, week-long training course. These RNs are prepared to provide specialized consultation and a unique supervisory level of clinical expertise in wound assessment and the specialized care involved for patients. Their training involves differentiation of wound types and the appropriate care; recognizing the effects of cancer, heart disease, diabetes, or COPD on wound healing; understanding care products and their implementation; wound-healing techniques; and patient education. (more…)
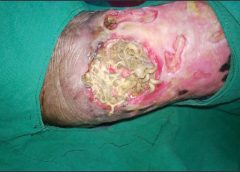
Read MoreCase study: Maggots help heal a difficult wound
Using maggots to treat wounds dates back to 1931 in this country. Until the advent of antibiotics in the 1940s, maggots were used routinely. In the 1980s, interest in them revived due to the increasing emergence of antibiotic-resistant bacteria.
At Select Specialty Hospital Houston in Texas, we recently decided to try maggot therapy for a patient with a particularly difficult wound. In this case study, we share our experience. (more…)
Read MoreThe Buzz Report: A wound care clinician’s best friend
By Donna Sardina, RN, MHA, WCC, CWCMS, DWC, OMS
In 2014, more than 8,000 new articles related to wound healing were added to the PubMed online database and hundreds of new patents for topical wound formulations were filed. Staying up-to-date with the latest and greatest findings and products can be challenging. We all lead busy lives, and our demanding work schedules and home responsibilities can thwart our best intentions. Although we know it’s our responsibility to stay abreast of changes in our field, we may feel overwhelmed trying to make that happen. (more…)
Read MoreAn easy tool for tracking pressure ulcer data
By David L. Johnson, NHA, RAC-CT
As a senior quality improvement specialist with IPRO, the Quality Improvement Organization for New York State over the past 11 years, I’ve been tasked with helping skilled nursing facilities (SNFs) embrace the process of continuous quality improvement. A necessary component of this effort has been to collect, understand, and analyze timely and accurate data. This article discusses a free tool I developed to help SNFs track their data related to pressure ulcers and focus their quality improvement efforts for the greatest impact. (more…)
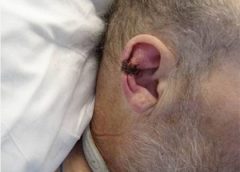
Read MoreUsing maggots in wound care: Part 2
By Ronald A. Sherman, MD; Sharon Mendez, RN, CWS; and Catherine McMillan, BA
Note From the Editor: This is the second of two articles on maggot therapy. The first article appeared in our July/August 2014 issue, Read part 1 here.
Whether your practice is an acute-care setting, a clinic, home care, or elsewhere, maggot debridement therapy (MDT) can prove to be a useful tool in wound care. But setting up any new program can meet resistance—and if you seek to establish a maggot therapy program, expect to meet significant resistance. By arming yourself in advance, you can achieve your goal more easily. This article covers all the bases to help you get your maggot therapy program off the ground. (more…)
Read MoreDevice–related pressure ulcers: Avoidable or not?
By: Donna Sardina, RN, MHA, WCC, CWCMS, DWC, OMS
A medical device–related pressure ulcer (MDRPU) is defined as a localized injury to the skin or underlying tissue resulting from sustained pressure caused by a medical device, such as a brace; splint; cast; respiratory mask or tubing; tracheostomy tube, collar, or strap; feeding tube; or a negative-pressure wound therapy device. The golden rule of pressure ulcer treatment is to identify the cause of pressure and remove it. Unfortunately, many of the medical devices are needed to sustain the patient’s life, so they can’t be removed. (more…)
Read MoreA Saudi rehabilitation facility fights pressure ulcers
By Joanne Aspiras Jovero, BSEd, BSN, RN; Hussam Al-Nusair, MSc Critical Care, ANP, RN; and Marilou Manarang, BSN, RN
A common problem in long-term care facilities, pressure ulcers are linked to prolonged hospitalization, pain, social isolation, sepsis, and death. This article explains how a Middle East rehabilitation facility battles pressure ulcers with the latest evidence-based practices, continual staff education, and policy and procedure updates. Sultan Bin Abdulaziz Humanitarian City (SBAHC) in Riyadh, Saudi Arabia, uses an interdisciplinary approach to address pressure-ulcer prevention and management. This article describes the programs, strategies, and preventive measures that have reduced pressure-ulcer incidence. (more…)
Read MoreNAWCO News
A letter from one of our own
I am writing this letter to share the sense of honor and privilege I felt in working with the National Alliance of Wound Care and Ostomy (NAWCO) to organize the first Eastern Region WCC Conference during the Fall of 2013. What an experience! In addition to representing a top-notch organization, I increased my knowledge of wound care and its products dramatically and met many amazing clinicians on the same journey.
Traveling from seven states and a wide variety of care settings, nearly 100 wound care clinicians came together with the common goal of enhancing their ability to make a difference in the lives of their patients. We launched the process of forming a vital clinician network that will allow us to share product and procedure information and experiences, leverage the information in our individual care settings, and strengthen the wound care knowledge of our peers.
Throughout this experience, I talked with dozens of local wound care product representatives. Like the clinicians, they provided a wealth of wound care insights. I encourage clinicians to reach out to these “best friends” of wound care and take advantage of their depth of knowledge.
I am passionate about advocating proper wound care, and if I can help disseminate wound treatment knowledge to other clinicians in support of their patients, I need no better reward.
Finally, the opportunity to represent NAWCO filled me with pride because of the respect I have for its vision. I believe in the importance of continuing education, furthering knowledge, and ensuring expertise through a certification process that is based on solid, research-based wound care. My hope is to represent NAWCO in the future.
Thank you for this exciting opportunity!
Sincerely,
Janie Hollenbach, RN, WCC, OMS,
DAPWCA, FACCWS
NAWCO names new executive director
The National Alliance of Wound Care and Ostomy (NAWCO), the largest wound care and ostomy certification organization in the United States, is pleased to announce that Cynthia (Cindy) Broadus, RN, BSHA, LNHA, CLNC, CHRM, WCC, DWC, OMS, was named executive director effective February 1, 2014.
Ms. Broadus brings a wealth of experience and knowledge to the organization. She has excelled for over 2 decades in specialty nursing care, litigation, corporate management, and company development. She joins the organization at a time of exciting change and will be instrumental in achieving accreditation that will provide national and international recognition for NAWCO.
“I am excited to be a part of such a great organization and supporting the efforts of the close-knit community that makes up the NAWCO certificants,” Ms. Broadus says.
Read the full press release.
DISCLAIMER: All clinical recommendations are intended to assist with determining the appropriate wound therapy for the patient. Responsibility for final decisions and actions related to care of specific patients shall remain the obligation of the institution, its staff, and the patients’ attending physicians. Nothing in this information shall be deemed to constitute the providing of medical care or the diagnosis of any medical condition. Individuals should contact their healthcare providers for medical-related information.
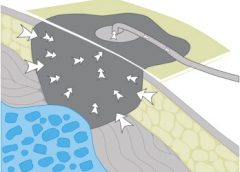
Read MoreGuidelines for safe negative-pressure wound therapy
By Ron Rock MSN, RN, ACNS-BC
Since its introduction almost 20 years ago, negative-pressure wound therapy (NPWT) has become a leading technology in the care and management of acute, chronic, dehisced, traumatic wounds; pressure ulcers; diabetic ulcers; orthopedic trauma; skin flaps; and grafts. NPWT applies controlled suction to a wound using a suction pump that delivers intermittent, continuous, or variable negative pressure evenly through a wound filler (foam or gauze). Drainage tubing adheres to an occlusive transparent dressing; drainage is removed through the tubing into a collection canister. NWPT increases local vascularity and oxygenation of the wound bed and reduces edema by removing wound fluid, exudate, and bacteria. (more…)
Read MoreClinician Resources: Intl Ostomy Assoc., Substance Use Disorder
Take a few minutes to check out this potpourri of resources.
International Ostomy Association
The International Ostomy Association is an association of regional ostomy associations that is committed to improving the lives of ostomates. Resources on the association’s website include:
- a variety of discussion groups
- information for patients
- list of helpful links.
The site also provides contact information for the regional associations. (more…)
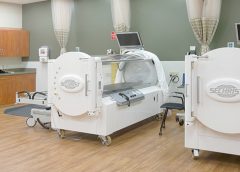
Read MoreMedicare reimbursement for hyperbaric oxygen therapy
By Carrie Carls, BSN, RN, CWOCN, CHRN, and Sherry Clayton, RHIA
In an atmosphere of changing reimbursement, it’s important to understand indications and utilization guidelines for healthcare services. Otherwise, facilities won’t receive appropriate reimbursement for provided services. This article focuses on Medicare reimbursement for hyperbaric oxygen therapy (HBOT). (See What is hyperbaric oxygen therapy?)
Indications and documentation requirements
Read More